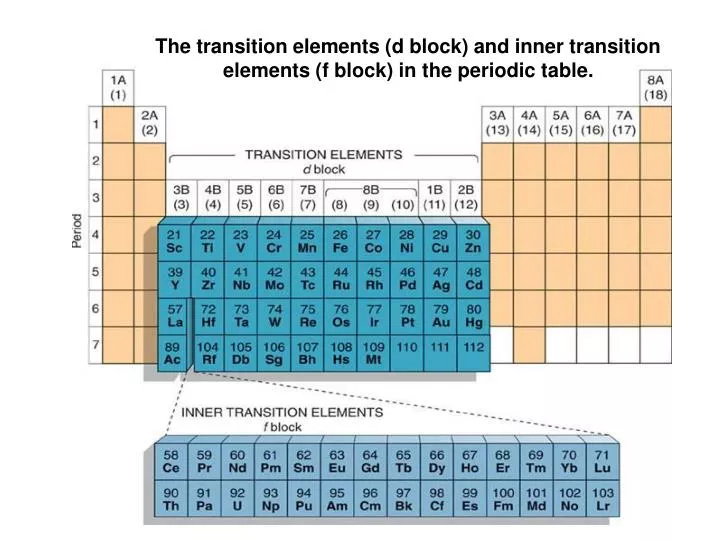
On going across the period melting points first increase, attain maximum value and then steadily decreases as atomic number.All the transition metals have high thermal as well as electrical conductivity and having very high melting and boiling points compared to those of representative elements due to their closed-packed structure.These are good conductors of electricity and heat. With the increase in atomic number, there is also an increase in the hardness of the metal. Metallic state- Lanthanide elements are generally metals.Generally, the oxidation state is +3 but it may be +4 and +2 due to the empty, fully filled, or partially filled f orbital. Oxidation state- The oxidation state of lanthanoid varies.When the 4f orbital is not shielded properly, the outermost electron gets affected by the positive nuclear charge, which causes lanthanoid contraction and is eventually responsible for the decreasing atomic radius. Atomic Radius- Along with the lanthanide series, there is a decrease in the atomic radius.In the elements of these series, the last electron enters the 4f orbital. These inner transition elements are not radioactive in nature, the only exception is Promethium. 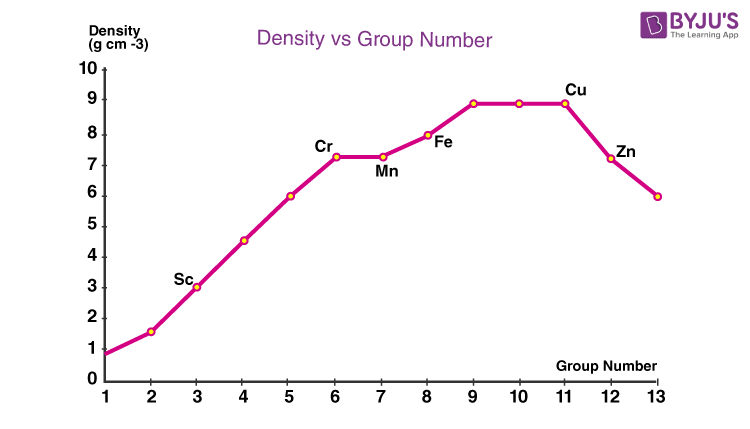
Examples-Įlements from atomic numbers 57 to 71 are included in the lanthanide series. They do not react and tend to be chemically inert. Their electrical conductivity is similar to other metals. These compounds have high melting points and tensile strength.These are called interstitial compounds- They do not have ionic, covalent, or non-stoichiometric characteristics.There are spaces in the transition metals where small atoms and molecules of hydrogen, carbon can get trapped.Examples- Bronze, Chromium steel, Solder, Stainless steel.Ĭheck How to Prepare for NEET 2022 Interstitial compounds of D block elements.The alloys formed have a high melting point and tensile strength in comparison to the initial characteristics of the metal. Such D block elements can form alloys.Due to the small difference between transitions element’s atomic radii, solid solutions can be formed when such difference is within 15%. 
Transition elements like Cr, Cu, Ag, Au, and Hg have oxidation state 1. In lower oxidation states, ionic compounds are formed whereas, in high oxidation states, covalent bonds are formed. Thus different oxidation states are observed.The electrons in the s and- d orbital of transitions element can form ionic as well as covalent bonds. The losing or gaining of electrons to form a chemical bond is defined as the oxidation state.While diamagnetism is due to the presence of paired electrons, paramagnetism is due to the presence of unpaired electrons, When such unpaired electrons are combined they give rise to ferromagnetism. Ferromagnetic- When magnetic characteristics are acquired by a substance.Paramagnetic- The presence of unpaired electrons in d orbitals leads to the paramagnetic characteristic of d elements With the increase in the number of unpaired electrons, the paramagnetic characteristic also increases.The relationship between transition elements and magnetic field gives rise to three magnetic properties of D elements: The only element in this series that exists in a liquid state is Mercury. The lowest melting and boiling point in the series is of elements of Group 12, Zn, Hg, and Cd.Transition elements with high melting and boiling point are CR, Mo, and W while manganese a,d Technetium have low melting and boiling point due to their weak bonding.The melting point increases and then decreases due to a decrease in the atomic number. The high melting and boiling point of transition elements is high due to their strong bonding. 5d elements have higher I.E in comparison to 3d and 4d elements. Thus palladium has the highest Ionization Enthalpy. 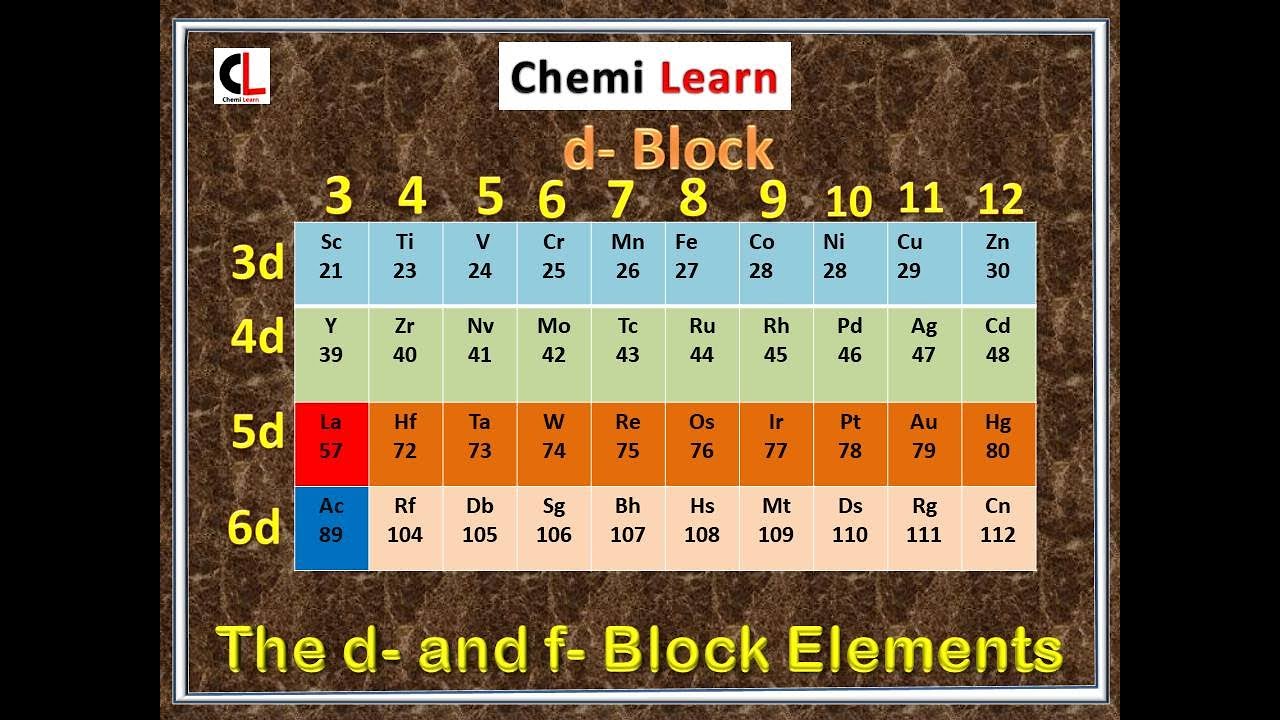
There are no electrons present in the s-shell.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
